Understand the Periodic Table and Element Properties
The periodic table is a cornerstone of chemistry, offering a structured way to understand the vast array of elements and their behaviors. Far from being just a list, it’s a powerful model that organizes elements based on their atomic structure, revealing predictable patterns in their properties. This guide will walk you through how to read the periodic table, understand the information it contains, and recognize the patterns that explain why certain elements behave similarly.
What You Will Learn
In this article, you will learn to:
- Identify the key information presented for each element on the periodic table.
- Distinguish between metals, non-metals, and metalloids and their general properties.
- Understand the significance of periods (rows) and groups (columns) on the table.
- Recognize the common properties of elements within specific groups, such as alkali metals, alkaline earth metals, halogens, and noble gases.
- Grasp the concept of valence electrons and the Octet Rule to explain element reactivity.
Prerequisites
- A basic understanding of atoms and their components (protons, electrons).
Understanding the Basics of the Periodic Table
Each element on the periodic table is represented by a box containing crucial information. Let’s break down what each part signifies:
1. Chemical Symbol
This is a shorthand, one- or two-letter abbreviation for the element. For example, ‘Fe’ represents Iron, ‘H’ represents Hydrogen, and ‘O’ represents Oxygen.
2. Atomic Number
The atomic number indicates the number of protons in the nucleus of an atom of that element. This number is fundamental because it uniquely identifies the element. For instance, Hydrogen has an atomic number of 1 (1 proton), while Oxygen has an atomic number of 8 (8 protons). The periodic table is arranged in order of increasing atomic number from left to right and top to bottom.
3. Atomic Mass
This number, usually found below the chemical symbol, represents the average mass of an element’s atoms. It’s an average because elements can exist in different forms called isotopes, which have different numbers of neutrons.
Classifying Elements: Metals, Non-metals, and Metalloids
The periodic table broadly categorizes elements into three main types based on their properties:
4. Metals
Metals constitute the majority of elements and are typically found on the left side of the periodic table. Their common characteristics include:
- Appearance: Usually shiny.
- State: Generally solids at room temperature (Mercury is a notable exception, being a liquid).
- Conductivity: Good conductors of heat and electricity.
- Malleability: Can be hammered into thin sheets.
- Ductility: Can be drawn into wires.
5. Non-metals
Found on the right side of the periodic table, non-metals exhibit very different properties:
- State: Can exist as solids, liquids, or gases.
- Appearance: Usually have a dull appearance.
- Conductivity: Poor conductors of heat and electricity.
- Solids: If solid, they tend to be brittle. (Diamond, an allotrope of Carbon, is a significant exception, being very hard).
6. Metalloids
Located in a zigzag region between metals and non-metals, metalloids share properties of both categories. They:
- May appear somewhat shiny.
- Are usually somewhat brittle.
- Act as semiconductors, conducting electricity better than non-metals but not as well as metals.
- Common examples include Boron, Silicon, and Germanium.
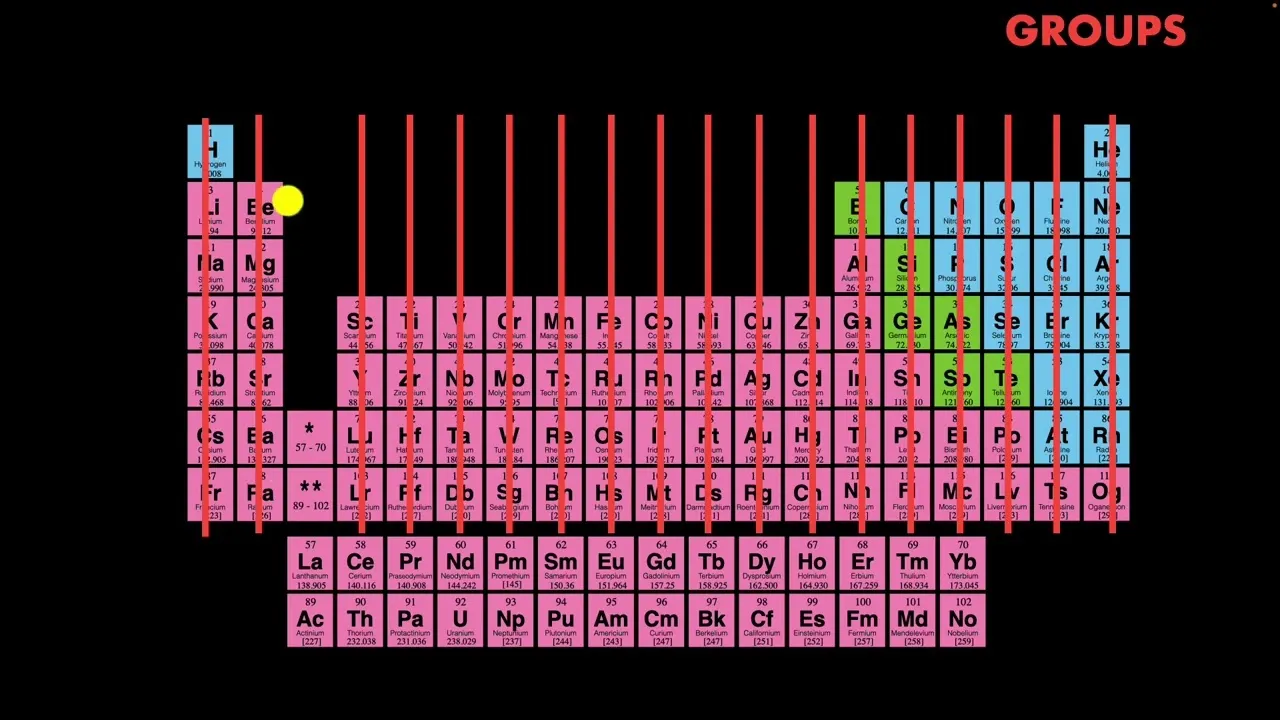
Understanding the Organization: Periods and Groups
The arrangement of elements on the periodic table is deliberate and reveals further patterns:
7. Periods (Rows)
The horizontal rows on the periodic table are called periods. There are seven periods. As you move from left to right across a period, the atomic number increases, and the properties of the elements gradually change, often transitioning from metallic to non-metallic characteristics.
8. Groups (Columns)
The vertical columns are known as groups. Elements within the same group share very similar chemical properties. This similarity is a key insight provided by the periodic table.
Groups can be labeled numerically from 1 to 18, or by an older system using Roman numerals and letters. Regardless of the labeling system, the grouping itself is what signifies similar behavior.
Key Groups and Their Properties
Certain groups on the periodic table are given specific names due to the characteristic properties of the elements they contain:
9. Alkali Metals (Group 1)
These include elements like Lithium (Li) and Sodium (Na). They are:
- Soft and silvery.
- Highly reactive, so much so that they are never found in their pure form in nature and are often stored under oil to prevent reaction with air or water.
- Note: Hydrogen (H), despite being in Group 1, is a non-metal and does not share these properties.
10. Alkaline Earth Metals (Group 2)
Examples include Magnesium (Mg) and Calcium (Ca). They are:
- Also reactive, but less so than alkali metals.
- Harder and denser than alkali metals.
- React more slowly with water compared to alkali metals.
11. Halogens (Group 17)
This group includes elements like Fluorine (F) and Chlorine (Cl). They are:
- Highly reactive non-metals.
- Rarely found in pure form; they typically combine with metals to form salts (e.g., NaCl – table salt).
12. Noble Gases (Group 18)
Examples are Helium (He) and Neon (Ne). These elements are distinct because they are:
- Almost completely unreactive, meaning they are very stable.
Why Elements Behave the Way They Do: Reactivity and Valence Electrons
The underlying reason for the patterns in reactivity lies in the arrangement of an atom’s electrons, particularly the outermost ones.
13. Valence Electrons
These are the electrons in the outermost energy shell of an atom. They play a crucial role in determining how an atom will interact with other atoms.
14. The Octet Rule
Atoms tend to react in ways that allow them to achieve a stable electron configuration, usually meaning having eight electrons in their outermost shell. This tendency is known as the Octet Rule.
- Noble Gases: Are stable because they already possess a full outer shell (2 electrons for Helium, 8 for others).
- Reactive Elements: Atoms with fewer than eight valence electrons will either lose, gain, or share electrons to achieve this stable configuration. For example, Sodium (Na) has one valence electron and tends to lose it to achieve the stable configuration of the noble gas before it. Chlorine (Cl) has seven valence electrons and tends to gain one electron to achieve the stable configuration of the noble gas after it.
15. Predicting Valence Electrons from Groups
A remarkable feature of the periodic table is that you can determine the number of valence electrons simply by looking at the group number:
- Group 1 elements have 1 valence electron.
- Group 2 elements have 2 valence electrons.
- Groups 13-18 generally have 3-8 valence electrons (Group 13 has 3, Group 14 has 4, …, Group 18 has 8).
- Exception: Helium (He) in Group 18 has only 2 valence electrons, but this is a full shell for it, making it stable.
This is why elements in the same group behave similarly: they have the same number of valence electrons, leading them to participate in chemical reactions in comparable ways.
Conclusion
The periodic table is far more than a chart; it’s an indispensable map of the elements. By understanding its structure—the atomic numbers, the organization into periods and groups, and the concept of valence electrons—you gain the ability to predict and comprehend the chemical behavior of elements and the compounds they form. This knowledge is fundamental to understanding the matter that makes up our world.
Source: The periodic table | Chemistry | Khan Academy (YouTube)